Protein Shapes Matter in Alzheimer’s Research
Often proteins misfold. When that transpires in the human mind, the pileup of misfolded
proteins can guide to neurodegenerative ailments like Alzheimer’s, Parkinson’s and
ALS.
Proteins do not misbehave and misfold out of the blue. There is a fragile ecosystem
of biochemical interactions and environments that normally allow them twist, unfold,
refold and do their jobs as they’re intended to.
About the Researcher
Having said that, as scientists from Michigan Technological College take a look at in an article
released in ACS Chemical Neuroscience, even a small alter might trigger extended-term implications.
For amyloid beta peptides — thought of a main hallmark of Alzheimer’s condition — a
widespread chemical modification at a certain spot on the molecule has a butterfly
impact that prospects to protein misfolding, aggregation and mobile toxicity.
Acetylation and Amyloid Beta Toxicity
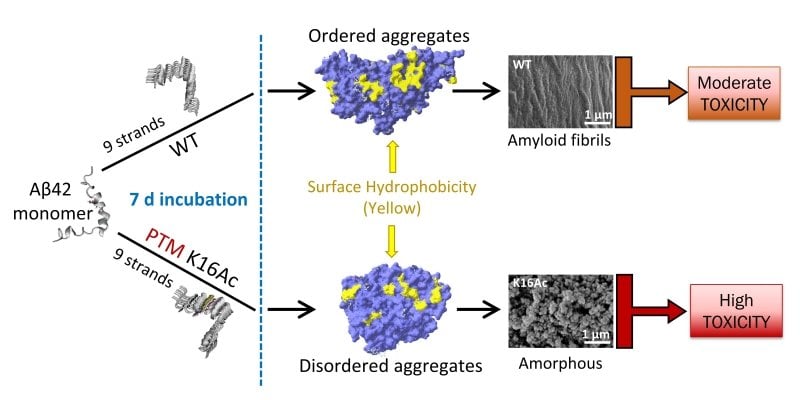
Ashutosh Tiwari, affiliate professor of chemistry at Michigan Tech, describes that misfolded amyloid beta proteins have a tendency to pile up and
form aggregates, which can form stringy fibrils or balled-up amorphous styles.
To fully grasp what leads to the various styles and to assess their toxicity, Tiwari’s
group looked at acetylation.


misfolded proteins building extended, stringy styles. Credit rating: Ashutosh Tiwari
Acetylation is a person of the most widespread chemical modifications proteins bear, but
a person of the the very least researched in terms of how it influences amyloid beta toxicity. On amyloid
beta proteins, acetylation can occur at two web-sites: lysine 16 and lysine 28.
The group uncovered that acetylation at lysine 16 led to the disordered aggregates that
shaped sticky but adaptable amorphous constructions and showed substantial ranges of toxicity.
They also uncovered the aggregates showed bigger no cost radical formation.
“No a person has accomplished a systematic examine to present if you acetylate amyloid beta it improvements
how the mixture seems, then it improvements its biophysical attributes and hence toxicity,”
Tiwari mentioned. “What we’re stating is that the shape, stickiness and versatility of the
aggregated protein framework can perform a essential function in the mobile toxicity and might
also have an affect on the system of toxicity.”
Aggregates and Alzheimer’s
In Alzheimer’s, these aggregates accumulate in the element of the mind that influences
memory. It is a condition that the Alzheimer’s Group stories is the sixth leading
trigger of dying in the US and will value the country about $305 billion in 2020. Tiwari
claims what we certainly want to fully grasp about the condition is that there is no one
trigger, no one trigger, and possibly no silver bullet mainly because of the chemistry concerned.
Grants and Funding
Analysis Excellence Fund, Michigan Technological College, Linda J. Horton Laboratory
Analysis Fund and Protein Misfolding Conditions Analysis Fund, Countrywide Institutes of
Wellbeing NIH R15 HL129213
“This is how a delicate alter on a one situation can have an affect on a entire protein’s aggregation,”
Tiwari mentioned, introducing that the impact of acetylation on tau, yet another protein aggregation,
has been far a lot more examined than amyloid beta. Also, quite a few scientists continue to consider a
misfolded protein has to appear a particular way to develop into problematic, and that other
misfolded varieties are much less of an difficulty.
Tiwari agrees some of the proteins’ improvements are delicate, and compares discerning the
differences and their consequences to snow tires. Snow tires have further treads and a a lot more
adaptable content to handle winter season roadways, but it is tricky to stage out these functions
at freeway speeds. Like various forms of tires, protein styles can surface indistinguishable
at a distance.
“This is not a little something that can be viewed from afar — it is a touch-and-come to feel residence,”
Tiwari mentioned. “We have to interrogate these attributes. We have to appear at these constructions
a lot more deeply from both morphology and biophysical perspectives.”
When we do, we might much better fully grasp the complexity of the misfolded proteins and
amyloid beta toxicity that can trigger neurodegenerative ailments like Alzheimer’s.
improvements in protein folding chemistry can trigger differences in shape and toxicity.
Credit rating: Ashutosh Tiwari
Michigan Technological College is a general public analysis college, home to a lot more than
seven,000 college students from fifty four international locations. Established in 1885, the College delivers a lot more than
120 undergraduate and graduate diploma systems in science and engineering, engineering,
forestry, business and economics, wellness professions, humanities, mathematics, and
social sciences. Our campus in Michigan’s Higher Peninsula overlooks the Keweenaw Waterway
and is just a handful of miles from Lake Outstanding.







