Solid-State Batteries Rev Up Electric Cars, Boost Grid Storage

Solid-state batteries promise to prove safer and longer lasting than conventional batteries. Now companies suggest they may commercialize solid-state batteries in the next five years for use in electric “hypercars” and power grids.
Conventional batteries supply electricity via chemical reactions between two electrodes, the anode and cathode, which typically interact through liquid or gel electrolytes. Solid-state batteries instead employ solid electrolytes such as ceramics.
Solid-state batteries can provide more energy than conventional batteries for the same amount of weight or space. “Solid-state batteries will be of huge benefit for electric vehicles, where the range is quite a key parameter,” says Noshin Omar, CEO and founder of Avesta Battery and Energy Engineering (ABEE) in Brussels. “Solid-state batteries are also much safer than conventional lithium-ion batteries, which use organic liquid electrolytes that are flammable and volatile.”
Now ABEE is helping develop a street-legal electric “hypercar,” the Fulminea, which is set to go to market in the second half of 2023. It will feature a hybrid battery pack that combines ABEE’s solid-state cells with ultra-capacitors.
“Currently our solid-state batteries have an energy density of about 400 watt-hours per kilogram, which is about double the typical energy density of commercial lithium-ion batteries on the market,” Omar says. “By 2025, we aim to achieve an energy density of 450 watt-hours per kilogram.”
“Metallic lithium has commonly been regarded as the holy grail of battery anodes. The silicon discovery opens up wide-ranging possibilities to adopt alternatives to metallic lithium.”
Italian automaker Automobili Estrema is developing the Fulminea, which will feature four electric motors with a total peak power of 1.5 megawatts (2,040 horsepower), allowing the car to accelerate from 0 to 320 km/h (200 mph) in under 10 seconds. Its 100 kilowatt-hour battery pack will give it an expected range of about 520 kilometers (323 miles).
ABEE is supplying the batteries, which use metallic lithium anodes, nickel-rich cathodes and a sulfide electrolyte, while battery specialist Imecar Elektronik will help package the cells. The battery pack will have a predicted weight under 300 kilograms, and Fulminea will have a predicted total curb weight of 1,500 kg.
“We are now aiming towards the scalability of our technology—optimizing the production process, the battery lifetime and the charging rate,” Omar says.
In addition, scientists at the University of California, San Diego, in partnership with electronics giant LG, have developed a new silicon all-solid-state battery that initial tests show is safe, long-lasting and energy-dense.
“With current trends, we aim to produce our first commercially relevant product by 2025, and achieve extensive market penetration by 2030,” says nanoengineer Zheng Chen at the University of California, San Diego.
Solid-state batteries with high energy densities have usually relied on metallic lithium for their anodes. However, these components place restrictions on battery charge rates and require warm temperatures during charging, usually 60 degrees C or higher. Silicon anodes can overcome these limitations, enabling much faster charge rates at room-to-low temperatures while maintaining high energy densities.
“Metallic lithium has commonly been regarded as the holy grail of battery anodes. The silicon discovery opens up wide-ranging possibilities to adopt alternatives to metallic lithium,” Chen says. “Additionally, silicon is a highly abundant, low-cost and safe material. This is a more environmentally friendly approach.”
![]()
1) The all solid-state battery consists of a cathode composite layer, a sulfide solid electrolyte layer, and a carbon free micro-silicon anode.
2) Before charging, discrete micro-scale silicon particles make up the energy dense anode. During battery charging, positive lithium ions move from the cathode to the anode, and a stable 2D interface is formed.
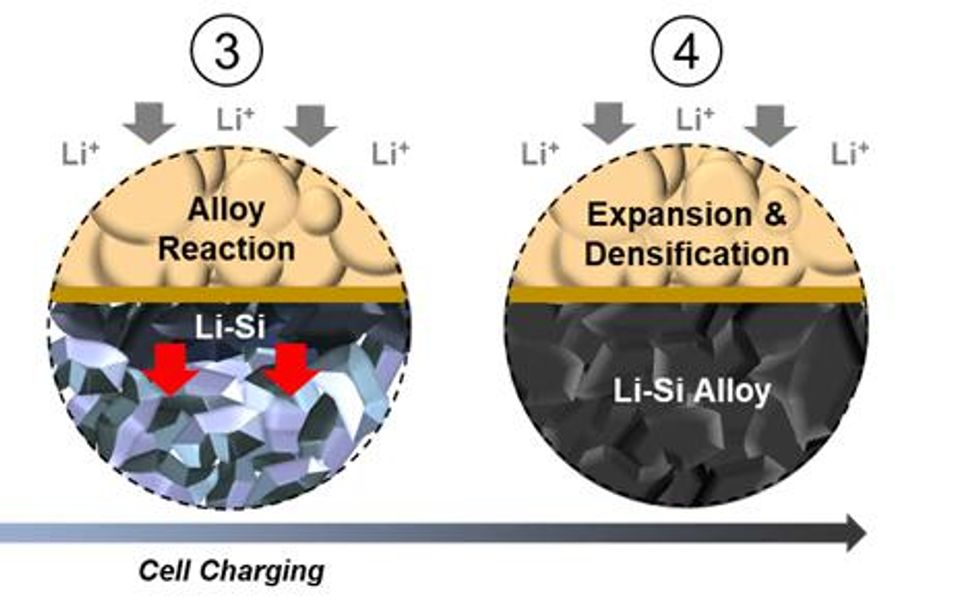
3) As more lithium ions move into the anode, it reacts with micro-silicon to form interconnected lithium-silicon alloy (Li-Si) particles. The reaction continues to propagate throughout the electrode.
4) The reaction causes expansion and densification of the micro-silicon particles, forming a dense Li-Si alloy electrode. The mechanical properties of the Li-Si alloy and the solid electrolyte have a crucial role in maintaining the integrity and contact along the 2D interfacial plane.
Scientists and battery makers have investigated silicon for decades as an energy-dense material to mix into, or completely replace, the graphite anodes seen in conventional lithium-ion batteries. In theory, silicon offers roughly 10 times the energy density of graphite.
However, previous attempts to add silicon to the anodes of lithium-ion batteries suffer from performance issues—specifically, the number of times such batteries can get discharged and recharged while maintaining performance is not high enough for commercial use. This is mostly because of how the silicon anodes could degrade when interacting with the liquid electrolytes they are paired with, as well as the way the silicon particles can greatly expand and contract in size as they recharge and discharge.
The new battery removed the liquid electrolyte, instead using solid sulfide-based electrolytes. These electrolytes were often believed to be highly unstable, but that was due to research on liquid systems that did not take into account the stability found in solid versions. The new study finds this electrolyte is extremely stable in batteries with all-silicon anodes.
“The proposed solid-state strategy overcomes the prevailing challenges associated with conventional liquid systems,” Chen says.
The scientists also removed all carbon and binders from the anodes. This significantly reduced the contact and unwanted side reactions they made with the solid electrolyte, avoiding continuous energy loss typically seen with liquid electrolytes. In addition, they used micron-scale silicon particles, which is less expensive than the nanometer-scale silicon particles often used in such work.
In tests, a laboratory prototype delivered 500 charge and discharge cycles with 80% capacity retention at room temperature. In contrast, previous studies with silicon anodes usually only achieved roughly 100 stable cycles.
The new batteries promise a high amount of energy density in terms of space. As such, the researchers suggest these devices could ultimately find use in grid storage applications.
“The silicon-based all-solid-state battery addresses the cost and safety concerns associated with conventional batteries for such applications,” Chen says. “If successful, every household will be equipped with energy storage systems powered by this innovation that lower their utility bills, provide a backup power supply, and support the global energy transition.”
In contrast, electric vehicles typically require batteries with a high energy densities in terms of weight. Still, “we are not ruling out automotive applications,” Chen says.
The scientists detailed their findings in the Sept. 24 issue of the journal Science. The university and LG Energy Solution have jointly filed a patent application on this work, and the university researchers have launched a startup, Unigrid Battery, that has licensed this technology.







