Graphene Jolts Sodium-Ion Battery Capacity
Following a long time of anticipation, sodium-ion batteries are commencing to produce on their guarantee for electricity storage. But so considerably, their commercialization is confined to big-scale utilizes this kind of as storing electricity on the grid. Sodium-ion batteries just never have the oomph required for EVs and laptops. At about 285 Wh/kg, lithium-ion batteries have two times the electricity density of sodium, earning them extra appropriate for people transportable apps.
Scientists now report a new form of graphene electrode that could increase the storage ability of sodium batteries to rival lithium’s. The material can pack almost as many sodium ions by volume as a traditional graphite electrode does lithium. It opens up a path to earning minimal-price, compact sodium batteries realistic.
Plentiful and low-priced, and with similar chemical homes as lithium, sodium is a promising substitute for lithium in upcoming-era batteries. The stability and basic safety of sodium batteries makes them specially promising for electronics and cars, wherever overheated lithium-ion batteries have occasionally tested harmful.
“But now the main challenge with sodium-ion batteries is that we never have a appropriate anode material,” says Jinhua Sunlight, a researcher in the office of industrial and elements science at Chalmers University of Engineering.
For the battery to demand immediately and retail store a good deal of electricity, ions want to quickly slip in and out of the anode material. Sodium-ion batteries use cathodes designed of sodium metallic oxides, although their anodes are ordinarily carbon-based anodes just like their lithium cousins though Santa Clara, California-based Natron Electricity is earning each its anodes and cathodes out of Prussian Blue pigment utilised in dyes and paints.
Some sodium battery builders are working with activated carbon for the anode, which holds sodium ions in its pores. “But you want to use superior-grade activated carbon, which is extremely expensive and not effortless to make,” Sunlight says.
Graphite, which is the anode material in lithium-ion batteries, is a reduce price option. On the other hand, sodium ions do not shift competently in between the stack of graphene sheets that make up graphite. Scientists utilised to feel this was since sodium ions are more substantial than lithium ions, but turns out even-more substantial potassium ions can shift in and out quickly in graphite, Sunlight says. “Now we feel it is really the floor chemistry of graphene layers and the digital framework that can’t accommodate sodium ions.”
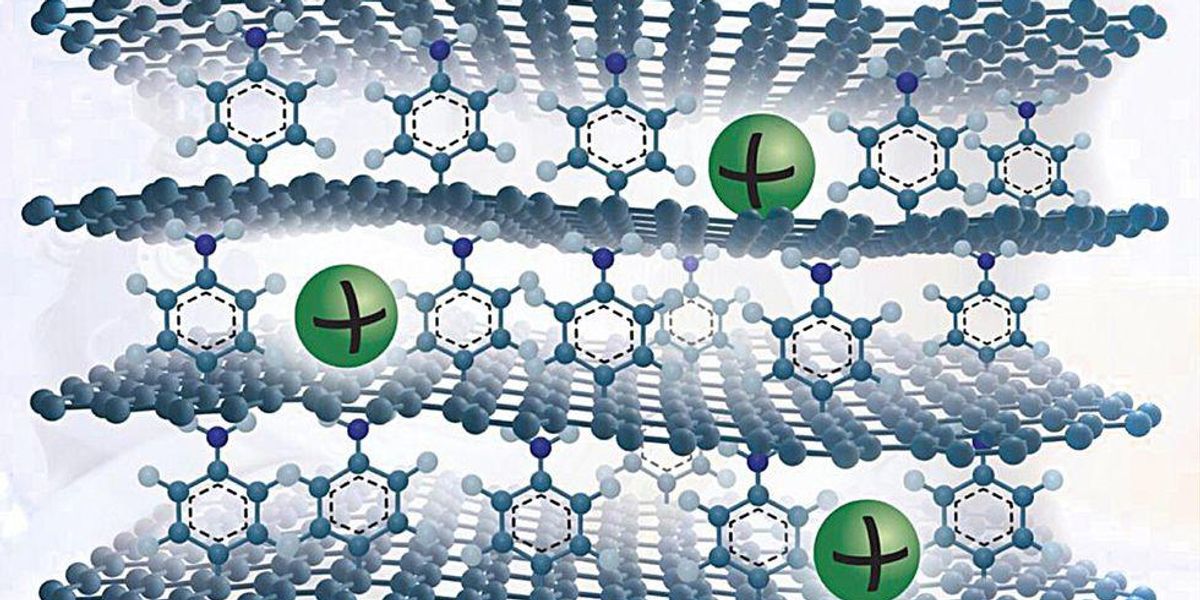
He and his colleagues have appear up with a new graphite-like material that overcomes these troubles. To make it, they grow a solitary sheet of graphene on copper foil and attach a solitary layer of benzene molecules to its prime floor. They grow many this kind of graphene sheets and stack them to make a layer cake of graphene held aside by benzene molecules.
The benzene layer improves the spacing in between the layers to enable sodium ions to enter and exit quickly. They also generate flaws on the graphene floor that as as active reaction internet sites to adsorb the ions. Additionally, benzene has chemical groups that bind strongly with sodium ions.
This seemingly uncomplicated approach boosts the material’s sodium ion-storing ability substantially. The researchers’ calculations show that the ability matches that of graphite’s ability for lithium. Graphite’s ability for sodium ions is ordinarily about 35 milliAmpere-hrs per gram, but the new material can hold in excess of 330 mAh/g, about the exact same as graphite’s lithium-storing ability.







